Randomized Controlled Trials | Vibepedia
Randomized controlled trials (RCTs) are a type of statistical experiment designed to evaluate the efficacy or safety of an intervention by minimizing bias…
Contents
Overview
Randomized controlled trials (RCTs) are a type of statistical experiment designed to evaluate the efficacy or safety of an intervention by minimizing bias through the random allocation of participants to one or more comparison groups. RCTs have been used to study a wide range of interventions, including drugs, surgical procedures, medical devices, and diets.
🎯 Origins & History
Origins paragraph — RCTs have their roots in the use of controlled experiments in medical research. Today, RCTs are a fundamental methodology in modern clinical trials, with applications in fields such as cancer research, cardiovascular disease, and infectious disease.
⚙️ How It Works
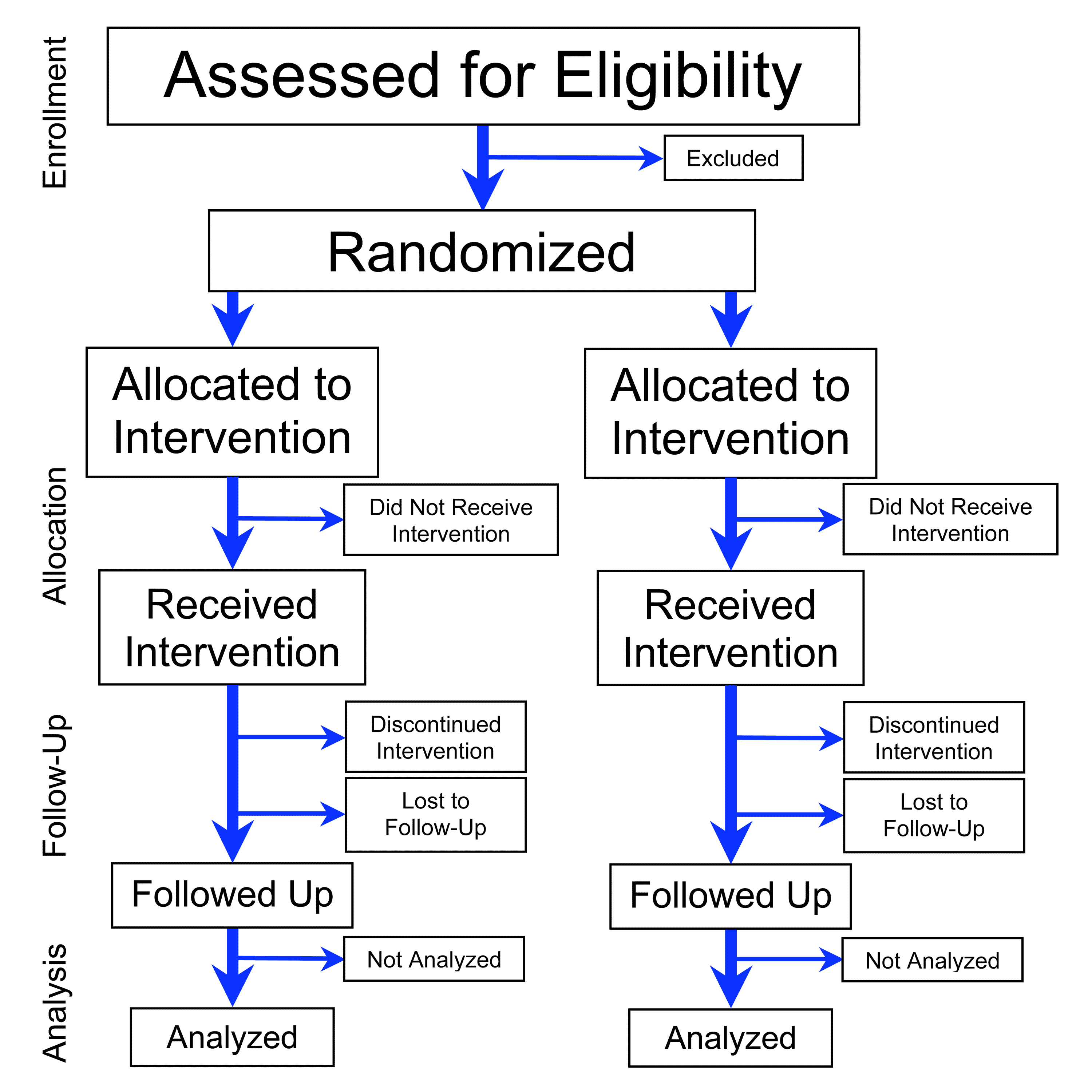
How it works — In an RCT, participants are randomly allocated to one or more comparison groups, with one group receiving the intervention under study and the other groups receiving an alternative treatment, a placebo, or standard care. This random allocation is designed to minimize bias and ensure that the groups are comparable in terms of known and unknown factors that could influence the outcome. The trial is typically conducted in a double-blind manner, where both the participants and the researchers are unaware of the group assignments. RCTs can be used to study a wide range of interventions, including pharmaceuticals, surgical procedures, medical devices, and dietary interventions.
📊 Key Facts & Numbers
Key facts — RCTs have been used to study a wide range of interventions. The COVID-19 pandemic has highlighted the need for more flexible and adaptive trial designs, such as platform trials and adaptive trials.
👥 Key People & Organizations
Key people — Some researchers have contributed to the development of RCTs.
🌍 Cultural Impact & Influence
Cultural impact — RCTs have been used to evaluate the effectiveness of interventions.
⚡ Current State & Latest Developments
Current state — The COVID-19 pandemic has highlighted the need for more flexible and adaptive trial designs, such as platform trials and adaptive trials.
🤔 Controversies & Debates
Controversies — RCTs have been criticized for their limitations, including the potential for bias in the selection of participants and the difficulty of generalizing results to real-world populations.
🔮 Future Outlook & Predictions
Future outlook — The future of RCTs is likely to involve the development of more flexible and adaptive trial designs. RCTs will need to be more inclusive, with a greater emphasis on diversity and representation in participant populations.
💡 Practical Applications
Practical applications — RCTs have a wide range of practical applications, including the evaluation of new interventions, the comparison of existing interventions, and the identification of factors that influence the effectiveness of interventions. RCTs can be used to study a wide range of interventions, including pharmaceuticals, surgical procedures, medical devices, and dietary interventions.
Key Facts
- Category
- science
- Type
- concept