Tissue Engineering Techniques | Vibepedia
Tissue engineering is a multidisciplinary field that combines cell biology, materials science, and engineering to create functional biological tissues for…
Contents
Overview
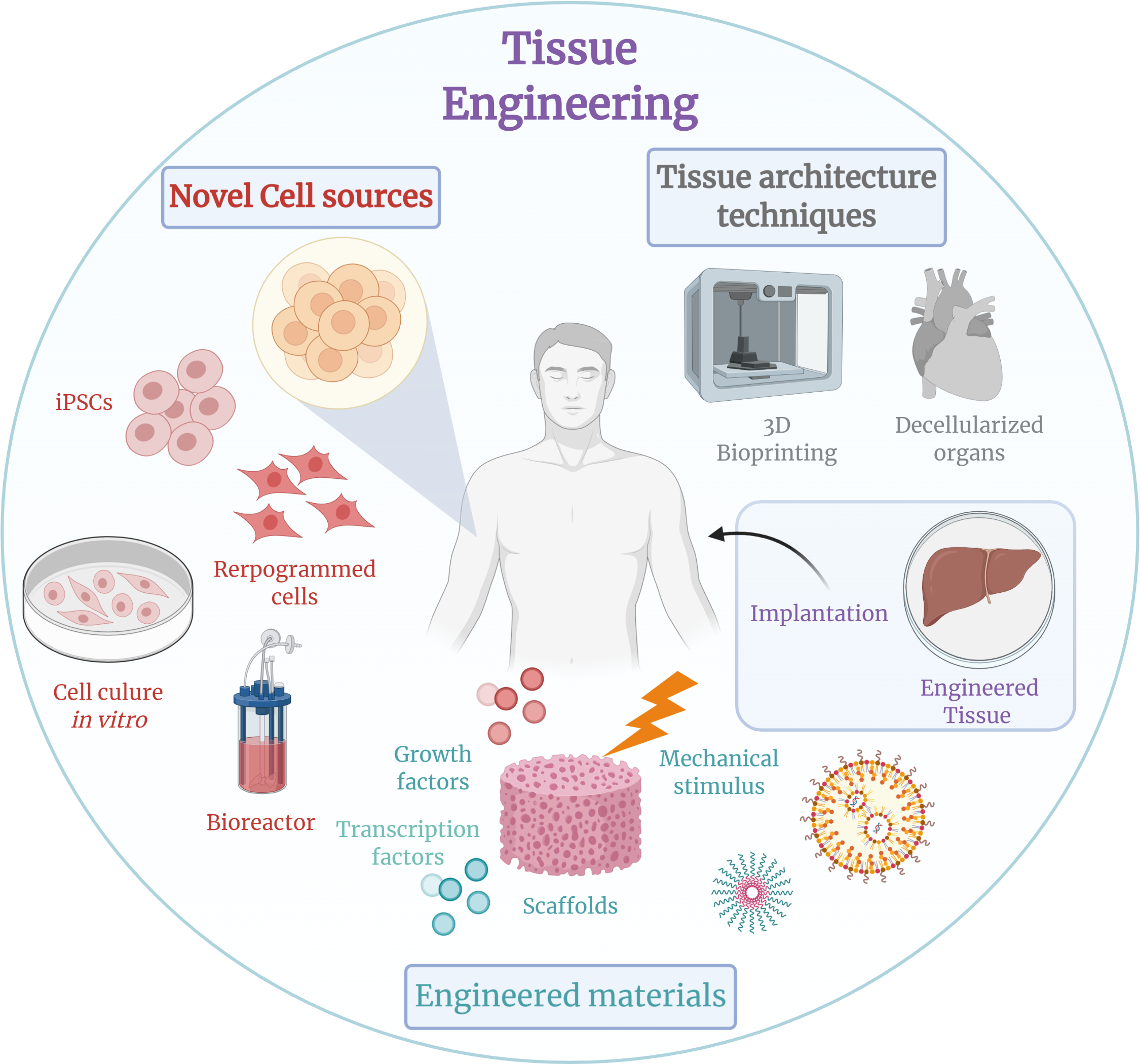
Tissue engineering is a multidisciplinary field that combines cell biology, materials science, and engineering to create functional biological tissues for therapeutic purposes. It aims to repair, replace, or regenerate damaged or diseased tissues and organs, offering potential solutions for a wide range of medical conditions. Key techniques involve the use of living cells, biocompatible scaffolds that provide structural support and guide tissue formation, and biochemical or mechanical stimuli to promote cell growth and differentiation. From skin grafts to complex organoids, these methods are pushing the boundaries of regenerative medicine, with significant advancements reported by institutions like the Wake Forest Institute for Regenerative Medicine and companies such as Organovo. The field grapples with challenges in vascularization, immune rejection, and scalability, but continues to evolve rapidly, promising a future where damaged tissues can be effectively restored.
🎵 Origins & History
The conceptual roots of tissue engineering stretch back to early attempts at skin grafting and organ transplantation. Early pioneers like Yale University's medical school researchers, including Joseph V. Bottino, explored the use of biomaterials to support cell growth. The term "tissue engineering" was popularized by researchers like Robert Langer and Joseph Vacanti. This period saw the emergence of foundational concepts like the use of scaffolds to guide tissue formation, moving beyond simple cell implantation to a more engineered approach.
⚙️ How It Works
At its core, tissue engineering involves three primary components: cells, scaffolds, and signaling molecules. Cells, often sourced from the patient (autologous) or a donor (allogeneic), provide the building blocks for new tissue. Scaffolds, typically made from natural polymers like collagen or synthetic materials like PLGA, offer a temporary structural framework that mimics the natural extracellular matrix. These scaffolds are designed with specific porosity and mechanical properties to encourage cell adhesion, proliferation, and differentiation. Signaling molecules, such as growth factors, are then introduced to direct cellular behavior and promote the formation of functional tissue, a process often accelerated in bioreactors that provide controlled environments.
📊 Key Facts & Numbers
The development of 3D-printed tissues has seen a dramatic increase, with over 50 companies now actively involved in this sub-sector. The cost of developing a single tissue-engineered product can range from tens to hundreds of millions of dollars, reflecting the complexity and extensive research required.
👥 Key People & Organizations
Key figures in tissue engineering include Robert Langer, a pioneer in biomaterials and drug delivery, and Joseph Vacanti, often credited with coining the term. Shinya Yamanaka's discovery of induced pluripotent stem cells (iPSCs) has revolutionized the field by providing a renewable source of patient-specific cells, earning him the Nobel Prize. Leading institutions like the Wake Forest Institute for Regenerative Medicine (WFI) and MIT's Koch Institute are at the forefront of research. Major companies like Organovo, Vericel, and Organogenesis are commercializing tissue-engineered products.
🌍 Cultural Impact & Influence
Tissue engineering has captured the public imagination, often appearing in science fiction as a pathway to immortality or effortless healing. Beyond speculative portrayals, its influence is tangible in the medical field, offering alternatives to traditional transplant surgeries and improving patient outcomes. The development of artificial skin for burn victims, for instance, has dramatically improved survival rates. Furthermore, the ethical discussions surrounding stem cell research and the creation of human tissues have spurred broader societal conversations about the boundaries of medical intervention and the definition of life itself, impacting public perception and regulatory frameworks.
⚡ Current State & Latest Developments
As of 2024, the field is witnessing rapid advancements in bioprinting complex vascularized tissues, moving beyond simple cell sheets to intricate 3D structures. Companies like BioLife Solutions are developing advanced preservation solutions for cell and tissue therapies, addressing critical logistical challenges. The integration of artificial intelligence and machine learning is accelerating scaffold design and predicting cell behavior, as demonstrated by research from Stanford University. Furthermore, there's a growing focus on developing off-the-shelf allogeneic therapies to overcome donor shortages and reduce immune rejection, with several products entering late-stage clinical trials.
🤔 Controversies & Debates
Significant controversies surround tissue engineering, particularly concerning the use of embryonic stem cells, which raises ethical objections for some. The potential for immune rejection of allogeneic tissues, despite advancements in immunosuppression and genetic engineering, remains a challenge. Questions about the long-term efficacy and safety of engineered tissues, especially for complex organs, are still debated. Furthermore, the high cost of these advanced therapies raises concerns about accessibility and equity, creating a potential divide between those who can afford regenerative treatments and those who cannot, a debate echoed in discussions around other advanced therapies.
🔮 Future Outlook & Predictions
The future of tissue engineering points towards the routine creation of complex, vascularized organs like kidneys and livers, potentially eliminating transplant waiting lists. Advances in gene editing technologies like CRISPR will likely play a crucial role in minimizing immune rejection and enhancing tissue function. We can anticipate the development of personalized tissue implants tailored to individual patient needs, moving beyond generic solutions. The integration of organ-on-a-chip platforms will also accelerate drug discovery and toxicity testing, reducing the reliance on animal models and providing more accurate human-relevant data, a trend championed by companies like Emulate Bio.
💡 Practical Applications
Practical applications of tissue engineering are already transforming healthcare. Skin grafts engineered from a patient's own cells are standard for treating severe burns and chronic wounds, with companies like Organogenesis leading in this area. Cartilage regeneration techniques are being used to repair damaged joints, offering alternatives to knee replacement surgery. Bladder reconstruction using engineered tissues has been successfully performed in clinical trials. Furthermore, engineered tissues are increasingly used in preclinical drug testing, providing more accurate models of human physiology than traditional cell cultures or animal models, a market segment actively pursued by firms like MatTek Corporation.
Key Facts
- Category
- science
- Type
- technology